
Are You Feeling the Pain of UDI Implementation?
The current regulations are the result of a final rule released in September of 2013 by the FDA, which stipulated that most medical devices carry a UDI. More regulations resulting from that ruling are scheduled to be rolled out through 2020.
The regulations include a number of significant benefits for both patients and manufacturers. They are also producing a number of challenges, some of which we touched on in a previous post.
We tapped into the insights of Kasey Tipping, ID Product Specialist of Cognex, to help us understand more about current issues with UDI implementation and how machine vision and barcode reading technology is used to solve these critical applications.
Listen to her interview here, and then read on for more insights into the UDI regulations.

What is a UDI?
“UDI” stands for Unique Device Identifier, and it’s part of a system for identifying medical devices through distribution and use. A UDI is a unique numeric or alphanumeric code. It must be displayed in both human readable (plain text) and machine readable (AIDC) form, and is comprised of a Device Identifier and a Product Identifier.
The UDI provides three distinct benefits:
1. Quick identification of devices
Every device will be required to provide a UDI. This allows your global supply chain to track a specific device throughout distribution. The regulations are prompting companies to integrate the tracking into their data systems.
2. Helps with recalls
In the event of a recall, you can pinpoint the specific device and quickly take action. In the event of an emergency, device information will be readily accessible, preventing medical errors.
3. Reduces or eliminates product counterfeiting
Because UDIs are tied to a global database, the threat of counterfeiters is dramatically reduced and the quality of overall medical services dramatically improves.
Device Identifier (DI) and Production Identifier (PI)
Each UDI is comprised of 2 parts:
1. Device Identifier
Includes the Global Trade Item Number and is specific to the device. It’s the fixed portion of the UDI, designed to identify the labeler and the specific version and model of the device.
2. Production Identifier
Includes a variety of production information. This is entirely up to the manufacturer, but it could include batch number, serial number, and expiration. This part of the UDI is much more conditional.
The DI and PI work together, much like the tracking and routing number on bank accounts. Here’s what they look like combined in a barcode:

It’s important to note, said Kasey Tipping of Cognex, that these two parts can be comprised of letters, numbers and characters. The most typical applications are a barcode, a QR code, or a matrix code.
“The labeler can decide how they want to do it,” Tipping said, “You want to make the decision that’s best for the product.” That typically involves using a code, which eliminates individuals reading off numbers and manually entering them into a database.
This video from the Registrar Corp (@registrarcorp) provides a great overview of DI and PI.
It’s noteworthy that the UDI must include a human-readable and machine-readable format — thus the numbers and the code.
Part of the Global Unique Device Identification Database
The Global Unique Device Identification Database (GUDID) is a global UDI database that will be maintained by the FDA in collaboration with the National Library of Medicine.
All UDI information will be stored in the database. It will be available to patients, caregivers, hospitals and the medical industry, and can be reached at the portal AccessGUDID.
Within that database, the UDIs will adhere to standards established by one of three FDA accredited issuing agencies — GS1 (@GS1Healthcare), HIBCC, and ICCBBA. Here’s a nice post on selecting your issuing agency (@meddeviceUDI, @lexisnexisIP).
UDI Compliance and Implementation Schedule
UDI implementation has been underway since 2014, with a series of rolling compliance dates occurring through 2020. The next big deadline is September 24, 2016 for Class II devices, and Direct Marking for Class III devices. (See Chart Below.)
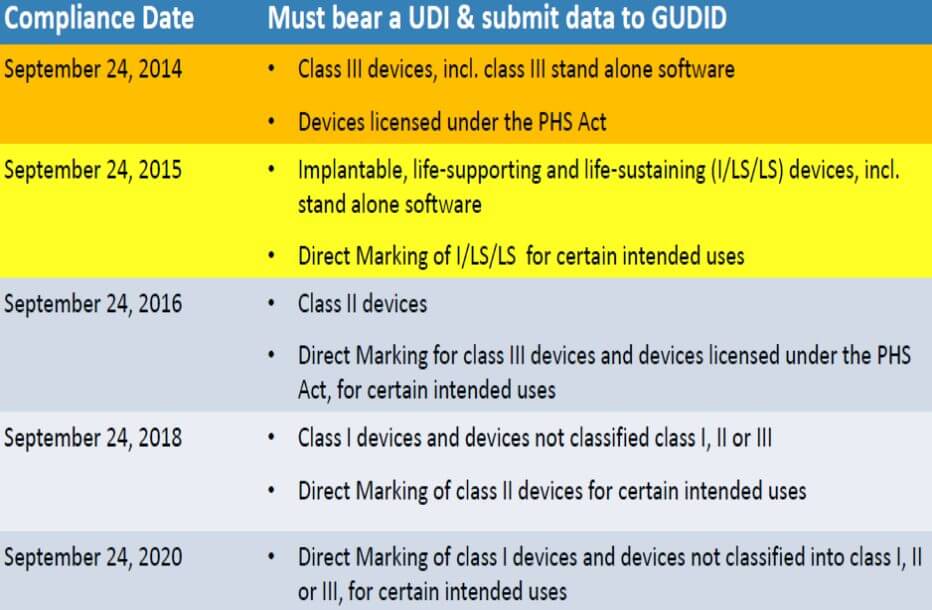
The FDA classifies medical devices in the following way:
Class I – Devices to be low risk and therefore subject to the least regulatory controls. (Examples: Colostomy bags, wound-care bandages)
Class II – Higher risk than Class 1, and require “reasonable assurance” of the device’s safety and effectiveness. (Example: Blood glucose test strips)
Class III – Highest risk — these are items that must be approved by the FDA. (Example: Surgical mesh)
Tipping notes, “Direct marketing for class III devices involves having a code on the part itself. That ensures it can be tracked, rather than having a code on the packaging that the part came in.”
Key Quality Issues: Verification, Validation and Mark Quality / Grading
Before we delve into some of the challenging areas for manufacturers, let’s take a look at three areas of concern: Verification, Validation and Mark Quality / Grading.
Verification
Verification is an industry standard that deals with the quality of the code, which includes how well the code is marked. It then assigns the code a grade (A-F).
According to Tipping, verification is typically done with 2D DataMatrix codes. “It inspects for things like contrast and finder/clocking patterns,” she said. Verification does not grade based on the encoded data, only the quality of the code. It requires strict lighting and positioning of the code to comply with industry standards.

Validation
Validation is another industry standard that focuses on the encoded data. It ensures that the data is formatted correctly and everything is in place to meet the regulation standard; for example, the DI and PI as previously mentioned.
Process Control Metrics / Mark Quality Assessment
Unlike Verification and Validation, which must meet industry standards, Process Control Metrics, also referred to as Mark Quality Metrics (as you might have gathered from the name) are an inline process used for quality control.
Unlike verification, the lighting and positioning are based on the application. They don’t have to adhere to a standard. It’s used to control the process. “For example,” Tipping said, “if the printer marking the codes is running out of ink, they will use metrics to track the contrast of the code.”
The Aches and Pains of UDI
We’ve touched on the potential UDI issues for manufacturers before. Let’s take a look at the some of the real-world issues that we are seeing today:
1. The rules are still in flux
The regulations are new, and because of that, unexpected issues are emerging — things that the FDA didn’t anticipate when they created the legislation. Manufacturers are beginning to encounter these challenges, and so we’ll see how the FDA responds.
2. Tiny codes, big problems
For medical devices that don’t allow much room for a code (think about a scalpel), getting the code placed on the product can prove to be difficult. Laser etching onto metals is the typical approach, but it’s not easy. The laser must be calibrated correctly, and the code must be marked clearly enough to be read.
3. Readability issues

A whole host of readability issues present themselves with UDIs. The Cognex graphic above shows the variety of issues that can affect the UDI reading, which includes:
- Washed out
- Blurring
- Low contrast
- Poor focus
- Finder degradation
- Uneven lighting
- Damaged and warped printing
- Badly printed
- Curved surfaces
- Faded
- Scratched
- Specularity
- Noisy background
- Thick printing
- Extreme perspective
4. UDI standards – implementation dates
A big issue is just having a thorough knowledge of UDI standards and the implementation dates. Is the data encoded properly to meet the standards? How do you know if a code is considered good or bad? Simply understanding the ins and outs of UDI implementation as a whole — whether it’s you or a strategic partner — can hold you up.
5. Not providing an adequate timeframe
The UDI regulations are coming fast, and your company shouldn’t assume you have enough time to implement and meet the regulations. It’s better to be too early than too late, so set your implementation process in motion. Or, if you don’t have one, start building one asap.
Seeking the UDI Solution
There’s no quick fix to these issues, but you can reduce your risk if you take the following steps:
1. Learn from FDA and GS1
Your first stop is to educate yourself on the basics of UDI and the Standards. Here’s a link to the FDA website, as well as the GS1 Quick Start Guide. It’s important you learn to walk before you run.
2. Consult with companies that understand industry standards
You either attempt to navigate through the industry standards yourself, or lean heavily on a company that understands the FDA regulations and the standards from GS1 for barcode reading, partnering with a company like Cognex that can provide applications for UDI.
The UDI regulations will continue to be rolled out through 2020, even though current guidelines are already in place. Don’t delay your preparation for the upcoming deadlines. Start making plans to ensure you’re in compliance today.


